In order to thrive in a changing climate, plants must adapt their physiology. These adjustments are not just about individual traits changing in isolation, plants must balance multiple processes at once, from carbon uptake to water loss and nutrient investment.
A new study led by LEMONTREE PhD student Astrid Odé (Utrecht University), published in Plant Ecophysiology, tests a key prediction from eco-evolutionary optimality (EEO) theory: that plant traits respond to environmental change in a coordinated way to maintain efficient resource use.
Coordination of Traits
Most studies of plant responses to elevated CO₂ have focused on single traits such as photosynthesis or stomatal conductance. But plants don’t operate trait by trait. Instead, they must balance carbon gain, water loss, and nutrient investment simultaneously.
The EEO theory that underpins the LEMONTREE research, offers a powerful framework for understanding this balance. It proposes that plants adjust their traits to maximise carbon gain while minimising the combined costs of water and nutrient use. In practice, this means that when one trait changes, others should shift in tandem to maintain overall efficiency.
Testing this idea experimentally, however, is not straightforward. It requires measuring multiple traits across different functional categories and determining whether their responses are coordinated.
A multi-trait experiment across crop species
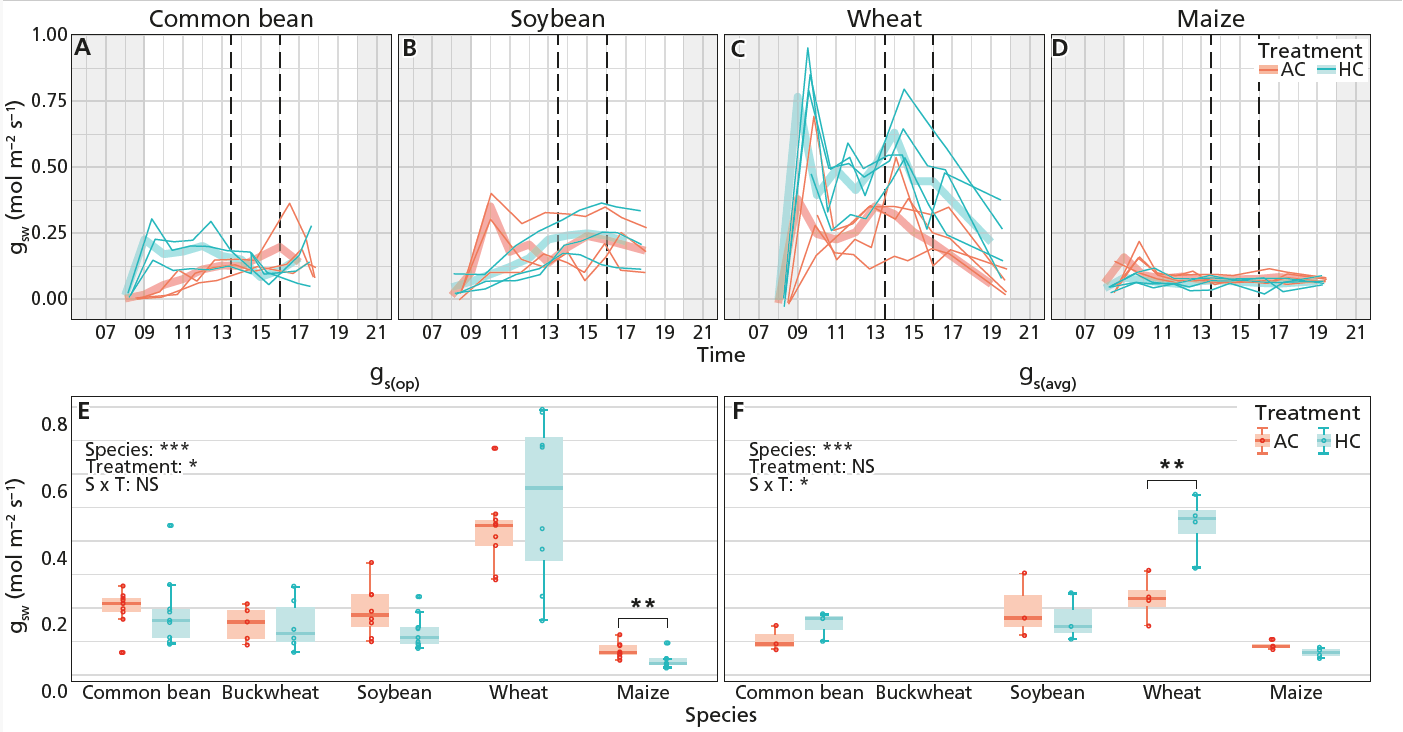
This study examined five widely grown crop species: four C3 plants—wheat, soybean, common bean, and buckwheat—and one C4 plant, maize. Plants were grown under controlled conditions at ambient (400 ppm) and elevated (1000 ppm) CO₂.
We measured a wide range of leaf traits, spanning:
- photosynthesis,
- photosynthetic biochemistry,
- stomatal conductance (including diurnal patterns),
- leaf morphology, and
- carbon and nitrogen content.
This comprehensive approach allowed us to look beyond individual responses and instead ask: do these traits move together in predictable ways?
A consistent pattern across C3 crops
Across the four C3 species, a clear and consistent pattern emerged.
Photosynthetic rates generally increased under elevated CO₂. At the same time, key biochemical traits, particularly the maximum rate of carboxylation (Vcmax) declined. Leaf nitrogen content also decreased, and stomatal conductance tended to be lower.
At first glance, this might seem contradictory: why would plants reduce investment in photosynthetic machinery while increasing photosynthesis?
EEO theory provides the answer. Under higher CO₂, carbon becomes less limiting, so plants can achieve similar or higher photosynthetic rates with less investment in costly enzymes like Rubisco. This frees up nitrogen for other uses and reduces unnecessary resource expenditure.
Crucially, these changes did not occur independently. The ratio between electron transport and carboxylation capacity shifted in a way that maintained a balance between the two processes. This aligns with the “coordination hypothesis,” which predicts that plants avoid overinvesting in any one component of the photosynthetic system.
In short, the plants were not just responding, they were optimising.
C3 stomata: dynamic but predictable
Stomata regulate both CO₂ uptake and water loss, making them central to plant efficiency. The study found that stomatal conductance generally declined under elevated CO₂, consistent with theory. Importantly, when we compared traditional midday measurements with diurnal averages, both captured similar responses. This suggests that widely used measurement approaches remain valid for testing optimality-based predictions. Which is great news, as the widely used midday approach is much less time intensive than measuring diurnal stomatal conductance.
Interestingly, anatomical limits on stomata showed little response over the short duration of the experiment. This highlights an important distinction: some traits can adjust rapidly (physiological responses), while others may require longer timescales (structural changes such as changes to the guard cells of stomata). It could also be that the plants responded also with other factors in the growth rooms, leading to net effects of no change in stomatal morphology, e.g. they also strongly respond to light availability and humidity, so that may have had an interactive effect together with CO2.

Two axes of C3 trait responses
The strongest test of EEO theory came from analysing how traits changed together. By applying principal component analysis (PCA) to trait response ratios, we identified clear patterns of coordination.
Two main axes emerged.
-
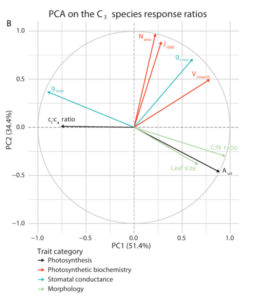
Figure 2. PCA analysis on the response ratios across the C3 species. The x-axis is PC1 and the y-axis is PC2. Arrows are eigenvectors per trait values with their corresponding loading and direction. - The first captured traits associated with the leaf economics spectrum, including photosynthesis, nitrogen content, stomatal conductance, and leaf size. This axis reflects how plants balance fast versus conservative resource-use strategies.
- The second axis grouped traits related to photosynthetic biochemistry and hydraulic capacity, linking carbon processing with water transport.
This partial decoupling suggests that while traits are coordinated, they are not rigidly locked together. Instead, plants retain some flexibility, allowing different combinations of traits to achieve similar overall outcomes. This partial decoupling may be key to enabling plants to adapt to a wide range of environments.
A different story for C4 maize
The inclusion of maize, a C4 species, highlights an important boundary for EEO theory.
Unlike the C3 crops, maize showed little change in photosynthetic biochemistry under elevated CO₂. This is expected: C4 plants already concentrate CO₂ internally, making them less sensitive to atmospheric increases.
However, maize still showed changes in photosynthesis and stomatal behaviour, likely linked to water-use efficiency rather than carbon limitation.
These differences reinforce the idea that optimality operates within biological constraints. The same principles apply, but the outcomes depend on the underlying photosynthetic pathway.
What does this mean for modelling and prediction?
One of the biggest challenges in predicting plant responses to climate change is representing how traits adjust over time. Many models rely on fixed relationships or empirical parameters, which can struggle to capture responses under novel conditions.
This study provides strong empirical support for an alternative: modelling based on optimality principles. By showing that multiple traits adjust in coordinated, predictable ways under elevated CO₂, the results validate a key assumption of EEO-based models like the P-model. Rather than prescribing how each trait should respond, these models allow optimal trait combinations to emerge from environmental conditions.
This is particularly important as CO₂ levels continue to rise. Understanding not just whether traits change, but how they change together, will be critical for predicting future carbon and water cycles.
While the study was in controlled conditions and focused on a small number of crop (which have a history of breeding and trait selection so may not be responding entirely similar to ‘wild’ plants), it opens the door to broader questions. How universal are these coordination patterns across ecosystems? How do they interact with other environmental factors like temperature and drought? And how do whole-plant traits fit into this picture?
What is clear is that plant responses to rising CO₂ cannot be understood in isolation. They are the result of integrated adjustments across multiple traits, shaped by the need to balance competing demands.
You can read the full paper here:
Ode, A., Drake, P.L., Veneklaas, E.J., Lankhorst, J.A., Rebel, K.T. & de Boer, H.J. (2026). Experimental evidence for coordinated leaf trait responses to elevated CO2 in five common crop species. Plant Ecophysiology, Doi: 10.53941/plantecophys.2026.100003